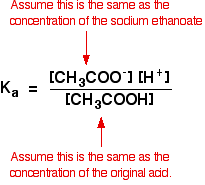
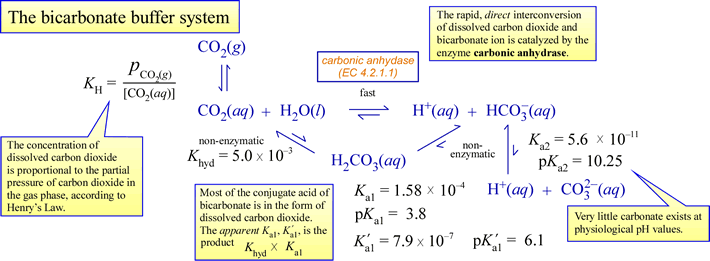
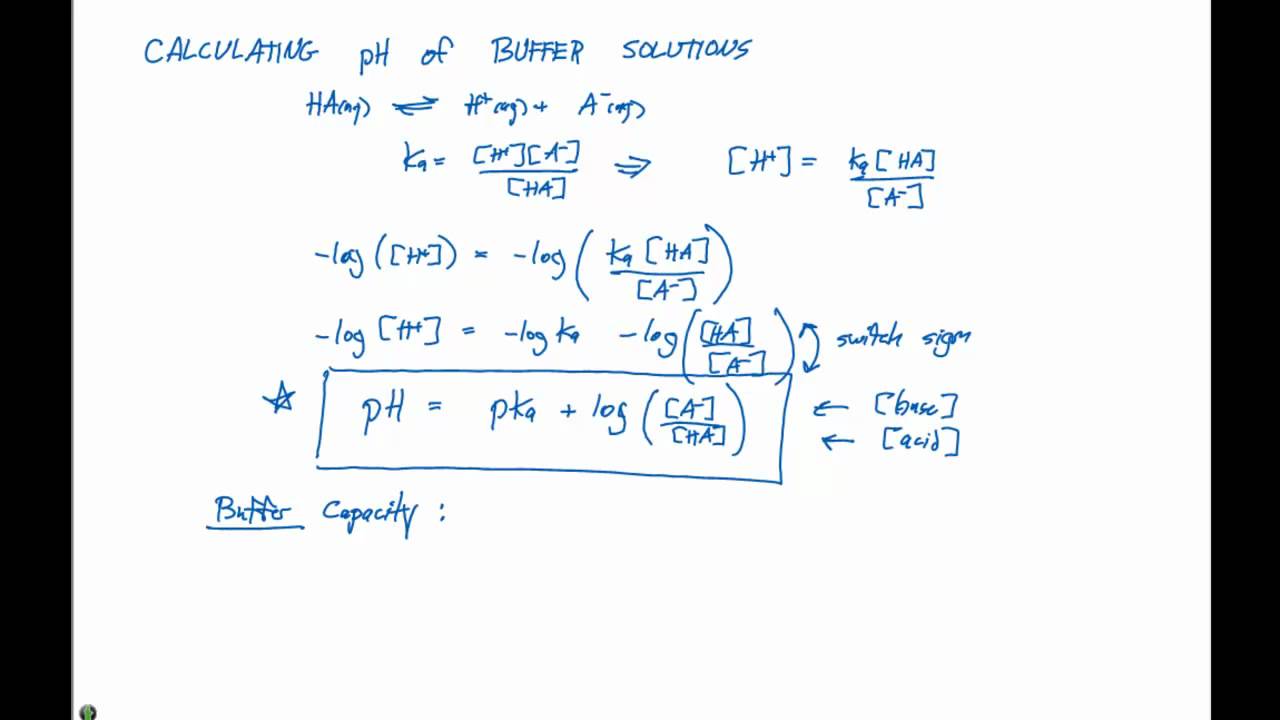
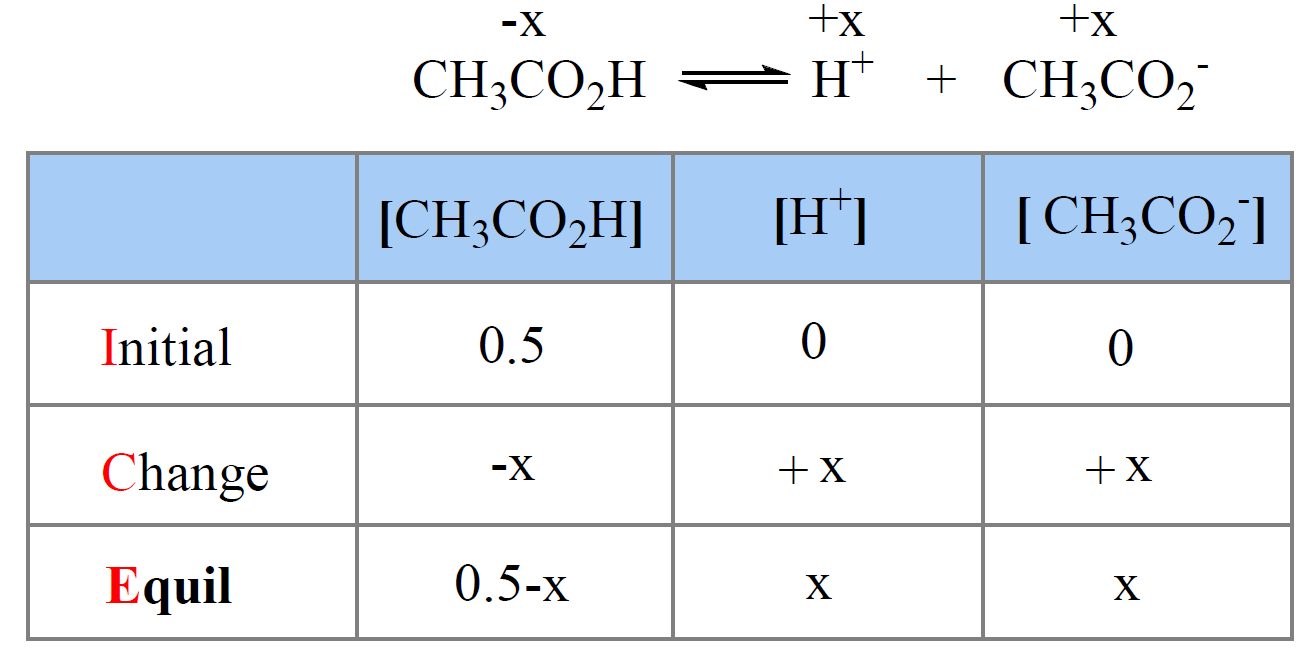
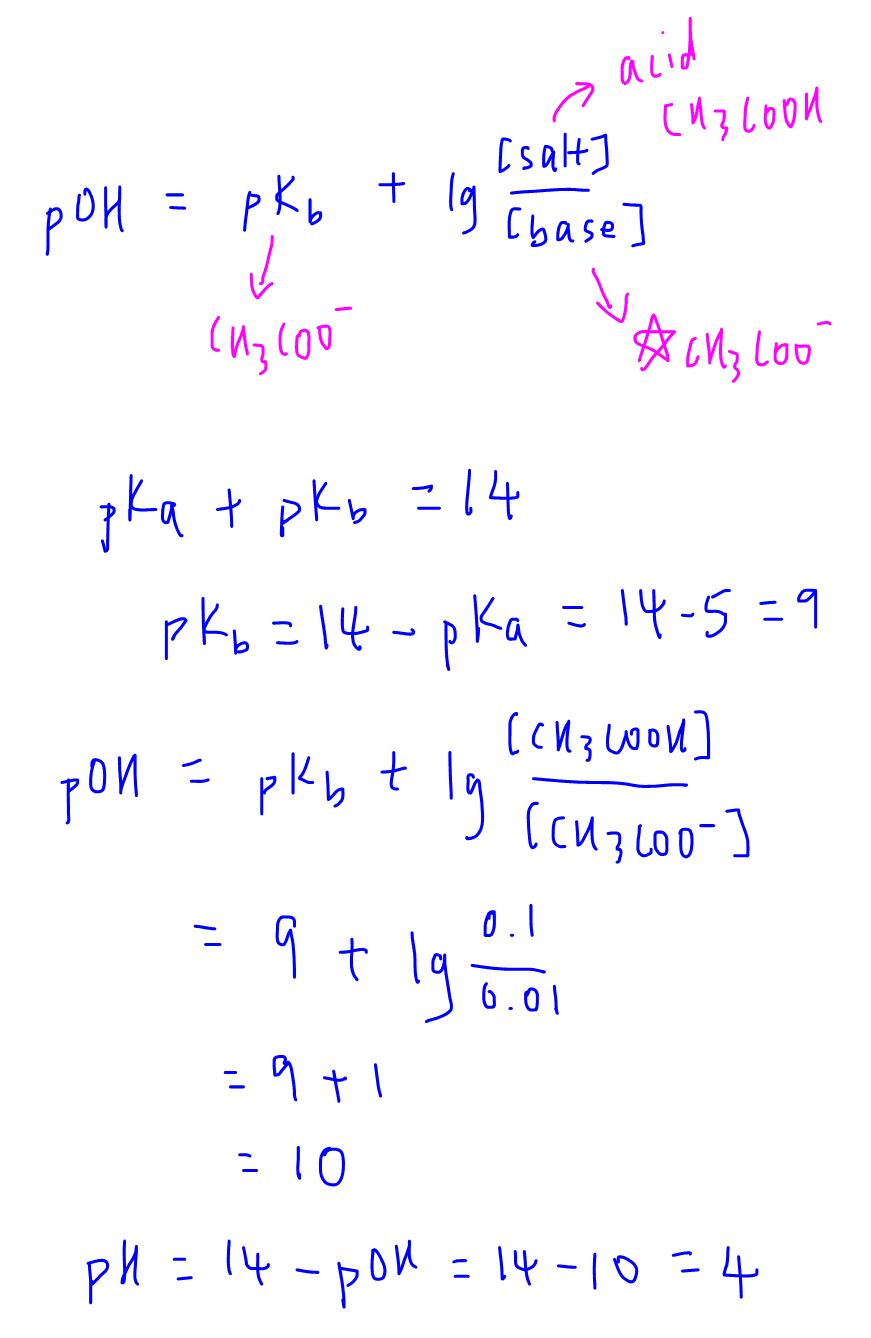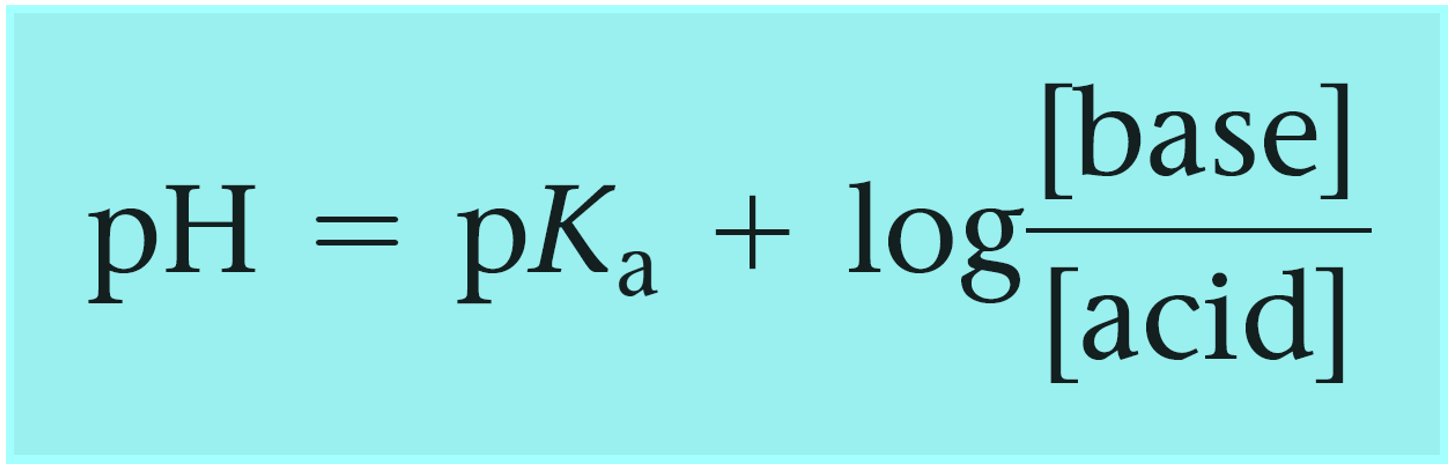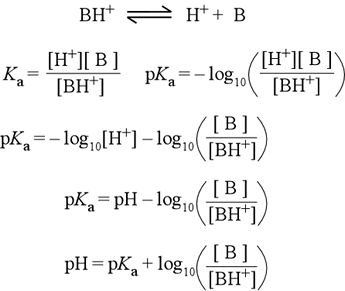How To Calculate the PH of a Buffer Solution | Equation & Example - Video & Lesson Transcript | Study.com

SOLVED: Use the Henderson-Hasselbalch equation to perform the following calculations. The Ka of acetic acid is 1.8 10-s Review your calculations with your instructor before preparing the buffer solutions Buffer A: calculate

SOLVED: B. pH of Buffer Solutions 9.50 g Mass of NaC2H3O2 * 3H2O (FW 136.5 g/mol) pH of Original buffer solution + HCl 4.5 pH of Buffer + NaOH 5.1 Mass of