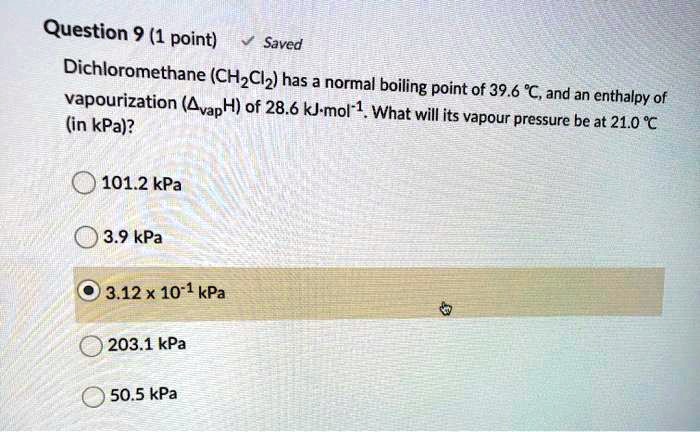
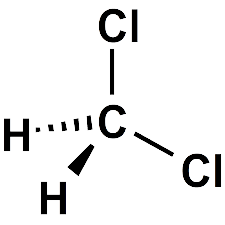
SOLVED: Water (H2O) and dichloromethane (CH2Cl2) are common solvents in a chemistry lab. Which of these solvents is expected to have a higher boiling point? Select one: a. Water because the molecules
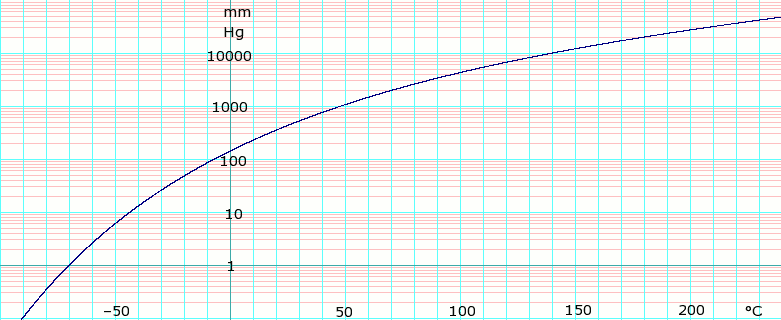
SOLVED: The vapor pressure of dichloromethane, CH2Cl2, at 0°C is 134 mmHg. The normal boiling point of dichloromethane is 40°C. Calculate its molar heat of vaporization. Express your answer to three significant

16.46 | Determine the normal boiling point (in kelvin) of dichloroethane, CH2Cl2. Find the actual - YouTube
Free Online Help: The vapor pressure of dichloromethane CH2Cl2 at 0`C in 134 mm Hg . The normal boiling point of dichloromethane is 40 `C . Calculate its molar heat of vapourization